After Scourfield (1940).

| Checklist of the Collembola: Note on the Morphology of the Prothorax |
Frans Janssens, Department of Biology, University of Antwerp, Antwerp, B-2020, Belgium
"Ici, la plupart des muscles longitudinaux dorsaux de la tête ne
sont pas relayés par des thoraciques, en raison de la réduction
du prothorax.
Une seule paire de muscles céphaliques passe au thorax et gagne le
mésonotum sans s'être attachée au foramen.
Tout sela prouve que la tête globuleuse est secondaire."
(Denis in Grassé, 1949:121 concerning the cervix ("constriction collaire") of Anurida).
Contrary to Carpentier (1949), who suggested a promesotergal fusion
in Entomobryomorpha, and more in line with Denis (1949), who presumed
that the globulose head capsule is a derived character, we hypothise that
the protocollembolan protergite was subdivided transversally
in a broad anteroprotergite and a narrow posteroprotergite.
The anteroprotergite fused with the posterior segment of the head capsule.
The remaining posteroprotergite lost its function as location of
muscular attachement in the 'new Collembola', reduced further,
and eventually also fused with the head capsule.
 |
An embryological perspective
Poduromorphan embryology:
Claypole (1898)
conducted a detailed study on the embryonic development of
Anurida maritma.
Claypole (1898:278) concludes that Anurida shows characters
allying it with crustaceans and myriapods
rather than insects .
Anurida is compared to a sexually matured insect embryo.
As a form that has lengthened its embryonic life
and has shortened its adult life.
Unfortunately, nothing is said about the prothoracic development.
Entomobryomorphan embryology:
Uemiya & Ando (1987,1991)
conducted a detailed study on the embryonic development of
Tomocerus ishibashii.
The embryonic development can be divided into nine stages.
During the sixth stage,
the dorsal region of the prothoracic segment begins to
develop at a slower rate than the mesothoracic and metathoracic segments.
The protergum becomes narrower and much less sclerotised,
one of the characteristic
features of Entomobryomorpha s.l. (Hopkin, 1997:144-145).
A chaetotactic perspective
In accordance with the regression of the prothorax,
the prothoracic chaetotaxy has been simplified (Cassagnau, 1974:302-303).
The dorsal chaetotaxy of the thoracic segments basically consists
of three rows of setae, 'a', 'm' and 'p', as proposed by Yosii (1956)
(André, 1988:514).
The naming of the last two cephalic rows of setae [in Xenylla] of da Gama,
namely cervicals (c) and parietals (p) is not generally followed.
Yosii (1956) proposed to name the most posterior row of the dorsal
cephalic setae, 'c', and the row immediately before, 'p'.
In 1960, Yosii changed his mind and reversed the two namings.
da Gama [(1980)] still follows the former terminology
while Cassagnau (1975) and others have adopted the latter
(André, 1988:508).
Thibaud (1970:134,142,144) in Ceratophysella bengtssoni (fig.10A,B),
in Schaefferia coeca (fig.11A,B), and
in Typhlogastrura balazuci (fig.12A,B)
describes the posterior region of the head as c-row and p-row setae
with respectively 4 and 7 setae,
and refers to the protergal setae as the medial row.
Cassagnau (1974:301[fig.1],302,303) describes
in his model of the cephalic chaetotaxy of the Poduromorpha
the occipital region of the head as c-row and p-row setae,
with respectively 4 and 5 setae,
and refers to the protergal setae as the anterior row or
posterior row, which he considers more evolutionary stable
than the medial row (1974:304).
André (1988:509[fig.2B],514[fig.6A]) describes
in the phanerotaxy of Xenylla jocquei
the posterior region of the head as c-row and p-row setae
and refers to the protergal setae as the anterior row.
Jordana & Arbea (1997:62[fig.23]) in Hypogastruridae
follow the original terminology of c-row and p-row of setae
at the posterior region of the head
and refer to the protergal setae as the medial row,
which is in-line with Yosii (Cassagnau 1974:304).
Fjellberg (1998:15,30[fig.10H]) describes
in the chaetotaxy of the Poduromorpha
the posterior region of the head as c-row and p-row setae
and refers to the protergal setae as the posterior row.
 |
Open issues:
What with seta p7 as sensillum?
In Onychiuridae: the posteroprotergite appears to bear more than just
the p-setae. Is this a secondarely derived character,
the result of polychaetosis?
Or are the Onychiuridae the most archaic poduromorphans,
and is the primitive chloting polychaetose and not homochaetose?
Are Onychiuridae an early side branch with a different,
larger posteroprotergite?
And/or desclerotisation between a-row and m-row in stead of between
m-row and p-row?
 |
In a generalised and simplified schema
(fig.m: threedimensional caudolateral reconstruction
of a longitudinal median section through the thoracic segments)
of the ancestral arthropodan thoracic musculature,
the following two basic categories of sets of muscles can be distinguished:
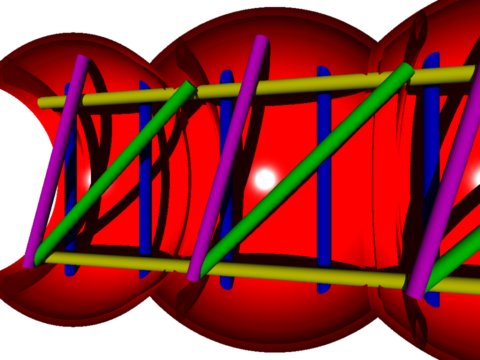 |
(Denis in Grassé, 1949:121) concerning the muscles of Anurida:
"Muscles tergo-tentoriaux. Les uns viennent du thorax et sont
souvent intersegmentaires avec deux innervations distinctes."
These muscles partly map
(Denis in Grassé, 1949:123) concerning the muscles of Anurida:
"Muscles longitudinaux dorsaux.
Il y a des longitudinaux dorsaux dans la région postérieure de la t�te;
ils appartiennent assurément au segment labial, mais il est impossible
d'affirmer qu'ils n'appartiennent qu'à lui, recevant leur innervation du
nerf r (fig.16) certainement complexe.
Le bord postérieur de la capsule céphalique répond è la limite thoraco-labiale
et c'est à son niveau que les longitudinaux dorsaux, céphaliques et
thoraciques, portent leurs plaques motrices.
Celles-ci résultent de la fusion des plaques motrices, destinées aux muscles
céphaliques, des nerfs r avec celles des nerfs intercalaires
(nerfs prothoraciques, tout au moins en partie) destinées aux muscles
thoraciques.
Cette jonction des nerfs atteste la condensation des domaines mitoyens
à deux tagmes."
These muscles map to the longitudinal protergal muscles of the
generalised schema, of which the anterior end is attached to the
protergal antecosta which is fused with the labial tergite
and of which the posterior end is attached to the mesotergal antecosta.
The generalised prothoracic muscular model can be mapped onto the muscular system of Orchesella and Neanura as described by Bretfeld (1963).
The following cephaloprothoracic muscles in Neanura (Schaller, 1970:30[fig.45b]) correspond subsequentially with the original prothoracic basic muscles:
 |
It is necessary to carefully reinterprete Bretfeld's schema of the
thoracic musculature of Neanura (Schaller, 1970:30[fig.45b]):
comparing the muscles of the labial segment, the prothorax and the
mesothorax shows the following analogy:
- the intersegmental muscles ism1-2 of the mesothorax
map to the muscle ism1b of the prothorax
- the dorsoventral muscles dvm2 of the mesothorax
map to the muscle ldvm1 of the labial segment
- the lateral dorsoventral muscles ldvm1-4 of the mesothorax
map to the muscles dvm1-3 of the prothorax
The protergal antecosta that is shown in Bretfeld's schema is
in fact an endoskeletal ridge of the posteroprotergite
to which are attached the lateral dorsoventral muscles
of the prothorax.
The following cephaloprothoracic muscles in Orchesella (Schaller, 1970:30[fig.45a]) correspond subsequentially with the original prothoracic basic muscles (see fig.x):
 |
 |
In the ancestral collembolan labioprothoracic segmentation,
where both the dorsal and the ventral primary intersegmental
areas of sclerotisation are continuous with the segmental sclerites following,
the typical arthropodan secondary segmentation prevails,
and the functional intersegmental rings
are the membranous posterior parts of the primary segments (fig. XX A, Mb).
The primitive intersegmental fold (Isg) forms an antecosta (Ac)
with externally an antecostal suture on both the
tergum (T) and the sternum (S),
and the precostal lip forms an acrotergite (atg) of the tergum
and an acrosternite (ast) of the sternum.
In a first phase,
the desclerotisation of a transvers protergal region
inbetween the medial row of setae and the posterior row of setae,
and assuming that the ventral sclerites retain the
form typical of secondary segmentation, the protergal sclerotisation takes the
form of an independent intertergite and eutergite.
The dorsal half of the labioprothoracic intersegmental membrane embraces the
intertergite (fig. XX B) and includes a part of the two adjoining
primary segmental regions.
The intertergite can be seen as the tergal antecosta having precostally
a narrow intertergal acrotergite
and postcostally a broad intertergal posttergite (ptg)
bearing the anterior and medial rows of setae.
In a second phase, the
intertergite is united with the labial tergum preceding (fig. XX C).
Since the intersternal
sclerotisation remains continuous with the prosternum following,
the labioprothoracic conjunctival membrane (Mb)
crosses obliquely on the side of the body ventrally
from the posterior part of the
labial segment to the anterior part of the prothoracic segment dorsally,
and the postcostal
intertergal posttergite becomes the posttergite of the labial segment.
The protergite of the ancestral protocollembolan is subdivided (fig.hp) into an anteroprotergite (B) that fused with the labiotergite to become an integrated part of the head capsule and a posteroprotergite (A) that becomes the 'protergite' of the extant Collembola.
The dipluran 'micronotum', as described by Verhoeff (1904)
(cited from Denis in Grassé, 1949:167), might be
homolog with our hypothetical intertergite.
The proturan 'gnathotergite', which is an isolated tergite in front of
their 'protergite' might also be homolog with the intertergite
(the anterior sclerite of the ancestral protergite).
So it seems that the origin of the reduction of the protergite goes even
further back in time.
Assuming that the current phylogenetic views on the 'close'
relationship between Collembola and Protura is correct,
the process of protergite reduction
seems to start even at the level of their common ancestor.
While in Protura the anterior part of the ancestral protergite remained an
independent sclerite and developed into the proturan gnathotergite,
in Collembola it fused with the posterior segment of the head.
The fact that proturan taxonomy uses a term as 'microcephalic' is also
interesting to note: it emphasises the difference between the not fused
protergite of the Protura (resulting in an apparently smaller head)
and the fused protergite
of the Collembola (resulting in an apparently larger head).
A morphometric perspective
Assume that the collembolan ancestor is derived from a
hypotethical arthropod with homogeneous segments and
generic locomotory appendages per segment.
Assume that the anterior segments already fused to form the head capsule.
While the abdominal appendages specialised functionally per segment,
to form the furca of the fourth abdominal segment,
the retinaculum of the third abdominal segment,
and the ventral tubus of the first abdominal segment,
the appendages of the second abdominal segment obliterated.
As a result of the reduction of the appendages of the second abdominal segment
the size of the sternum reduced proportionally.
Due to the sternal reduction of the second abdominal segment,
all anterior sterna are dislocated posteriorly.
This can be observed best in Poduromorpha and
Entomobryomorpha s.l.: in lateral view, the coxae appear to be intersegmental,
ventrodorsally aligned with the tergal conjuctivae.
Due to the posterior dislocation of the thoracic sterna, the head capsule
becomes slightly tilted in the downward direction.
Due to the downwards tilt of the head capsule,
the protergum and propleura are exposed to additional stress.
The additional stress in the cuticula is compensated
by the labioprotergal fusion as described above.
 |
| No | Function | Neanura | Orchesella |
|---|---|---|---|
| 1 | Primary levator | I dlm1 + dlm2 | Idlm |
| 2 | Lb dlm1 + dlm2 | (absent) | |
| 3 | I vlm | Ivlm | |
| 4 | Primary promotor | Lb vlm2 | Lbvlm2 |
| 5 | Secondary levator | (absent) | Idlm |
| 6 | Secondary promotor | Lb vlm1 | Lbvlm1 |
| 7 | Primary promotor | I ism1b | Ism3u4 |
| 8 | Primary levator | dvm2 | Lbldvm |
| 9 | Secondary levator | I ism1a | Iism1 + Iism2 |
Conclusion 1:
Due to the increased longitudinal stress at the tergum,
caused by the downwards tilted head capsule,
the function of the
primary tergal longitudinal muscle
from mesotergal antecosta to protergal antecosta (no. 1)
got enforced by replicating
a secondary tergal longitudinal muscle
from mesotergal antecosta to labiotergal antecosta (no. 5).
In Neanura this secondary muscle is (still) lacking.
In Orchesella it is fully developed.
Conclusion 2:
Due to the labioprotergal fusion,
the primary tergal longitudinal muscle
from protergal antecosta to labiotergal antecosta
(no. 2) lost its function, it became redundant and reduced.
In Neanura it is still available.
In Orchesella it is completely obliterated.
Both indicate that the Poduromorpha are phylogenetically more ancestral
then the Entomobryomorpha.
 |
Chaetotactic confirmation,
based on Synameria orientalis Cassagnau, 1990:21 Fig.1 :
1. the lateral macrosetae of the thoracic lobes:
note the consistency in the number and position of the two lateral
macrosetae on the lobe of the protergum and of the posterior lobe of
meso- and metatergum.
2. the number of setulae of the posterior row of the thoracic terga:
note the consistency in the number of setulae of the
posterior row (p-row): 4 setulae.
Both are an indication that the posterior part of the meso- and metatergum
is homolog with the protergum.
3. the number of setae of the mediodorsal region of the anterior row:
the mediodorsal region of the anterior row (a-row) of the
meso- and metatergum bears 4 setae;
the mediodorsal region of the 'anterior row' of the
posterior region of the head bears 4 setae.
4. the lateral cluster of anterior setae:
the lateral region of the anterior lobe of the
meso- and metatergum bears a cluster of three setulae and one macroseta;
the cephalic lobe
bears a cluster of three setulae and three macrosetae
(a result of plurichaetosis?).
Both are an indication that the anterior part of the meso- and metatergum
is homolog with the posterior tergal part of the head.
 |
The neanurid genera Caputanurina Lee, 1983 and Leenurina Najt & Weiner, 1992 (cited from Hopkin, 1997:30,31; cited from Deharveng, 2004:422,424; cited from Deharveng, Bedos & Weiner, 2011:40) are an extant example of the final completion of the evolutionary trend in the fusion of the occiputo-prothoracic tergites in Poduromorpha. Traditionally, in the Caputanurininae, the headcapsule "is fused with the prothoracic tergite". Stebaeve (1988) and Najt & Weiner (1992) consider this an extraordinary hyperevolution of a Pseudachorutinae lineage (cited from Deharveng, 2004:422), possibly via the very similar Pseudachorutinae (but with not fused protergite): Koreanurina Najt & Weiner, 1992 (cited from Deharveng, Bedos & Weiner, 2011:40), while Lee (1983) gave the genus subfamily status (Caputanurininae). These three genera represent different degrees in head-prothorax fusion, from separate in Koreanurina to completely fused in Leenurina, challenging the validity of the subfamily Caputanurininae as currently defined (cited from Deharveng, Bedos & Weiner, 2011:40).
We challenge this extraordinary hyperevolution of head-prothorax fusion
with our hypothesis of the more generalised head-acroprotergite fusion in
the Collembola and consider the Caputanurininae as living proof of the ultimate
evolutionary step in Poduromorpha by total reduction of the protergite.
They share this feature with the Neocollembola.
Therefore, the Caputanurininae can be considered as the extant descendants
of a lineage of Collembola inbetween Poduromorpha and Neocollembola.
In contrast, based on a comparison of the prothoracic musculature with a basic thoracic musculature model, we presume that the protergite and mesotergite are not or only partly fused, but that at least the mesotergal acrotergite enlarged. In any case, a clear tendency to enlarge the mesotergum can be observed. We will illustrate our presumption with two case studies: Isotomurus maculatus (Isotomidae), based on Hutasse-Jeannenot 1974:93, and Pogonognathellus longicornis (Tomoceridae), based on Carpentier, 1949:48,49.
 |
In our opinion, muscle (3) corresponds with
the oblique promeso-intersegmental muscle (green),
the ventral end attached to the prosternal apophyse/antecosta
and the dorsal end attached to the mesotergal antecosta
(see the basic thoracic musculature model of fig.m2).
Muscle (4) corresponds with
the intrasegmental dorsoventral mesotergosternal muscle (magenta),
the ventral end attached to the mesosternal apophyse/antecosta,
and the dorsal end attached to the middle of the mesotergite.
It is clear that the mesotergal antecosta is displaced posteriorly
and that the mesotergal acrotergite correspondingly is enlarged.
 |
To conclude: the enlargement of the mesotergum is mainly due the enlargement of the acrotergite of the mesotergite. This implies that the antecosta of the mesotergite must be displaced more posteriorly (see arrow in fig.2). The tendency to enlarge the meso-acrotergite reaches its climax in Lepidocyrtus in which, in some species, the meso-acrotergite forms a prominent cap that spans the head capsule, such as in Lepidocyrtus paradoxus.
In the context of our labioprotergal fusion hypothesis, two pathways for the enlargement of the acrotergite can be envisaged:
<-pC-+----------------- Poduromorpha
|
+--N-+------------ Neelipleona
|
+------------ Entomobryomorpha s.l.
|
+------------ Symphypleona
|